
The transition to freshwater by marine crustaceans certainly required the evolution of morphological, physiological, and behavioral adaptations for success in dilute media. Life in freshwater, unlike marine systems, is incredibly stochastic in the levels of major ions, oxygen, and pH. Accordingly, the biological innovations of early freshwater inhabitants to maintain osmotic and ionic gradients intracellularly and in their extracellular fluids are, by far, the cornerstone of their existence in a low salinity milieu. Amphipod crustaceans demonstrate superiority in these marine-freshwater transitional feats, as evidenced by their ubiquity in freshwater systems around the globe.
Some freshwater amphipod species, like Hyalella azteca, are significant and widely used crustacean models in ecotoxicology in North America. However, remarkably little is known about the basic osmoregulatory and respiratory physiology that dictates their tolerance thresholds and is critical to their success across a breadth of their ecological niches.
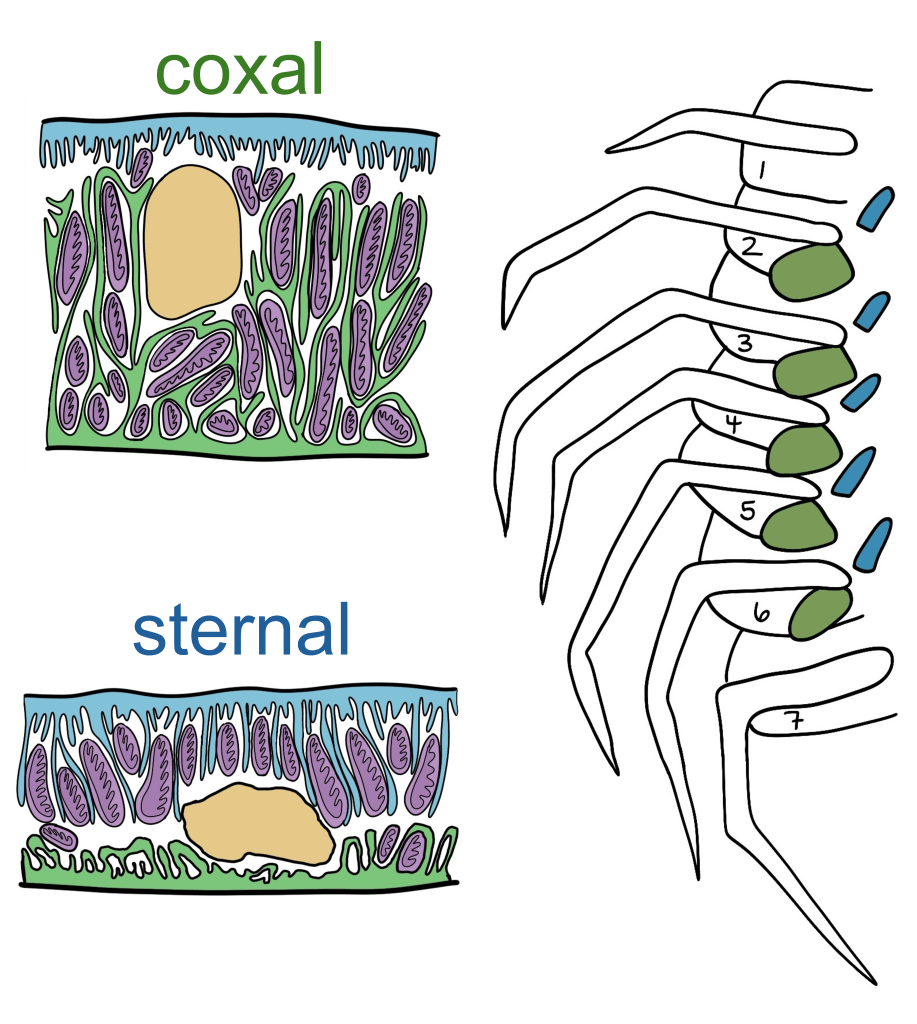
Our research has the primary goals of advancing the field by describing the cellular and organ-specific mechanisms of H. azteca to (1) attenuate passive ion loss and water gain, and (2) to tolerate a wide range of salinities and hypoxic environments. Freshwater amphipods have had to reorganize ion and water transport proteins and select new ones compared to their marine cousins, and members of the suborder Senticaudata (including H. azteca) have gone a morphological step further and evolved an additional set of gills. The regular large and pouchy coxal gills (green) and smaller, tubular sternal gills (blue)
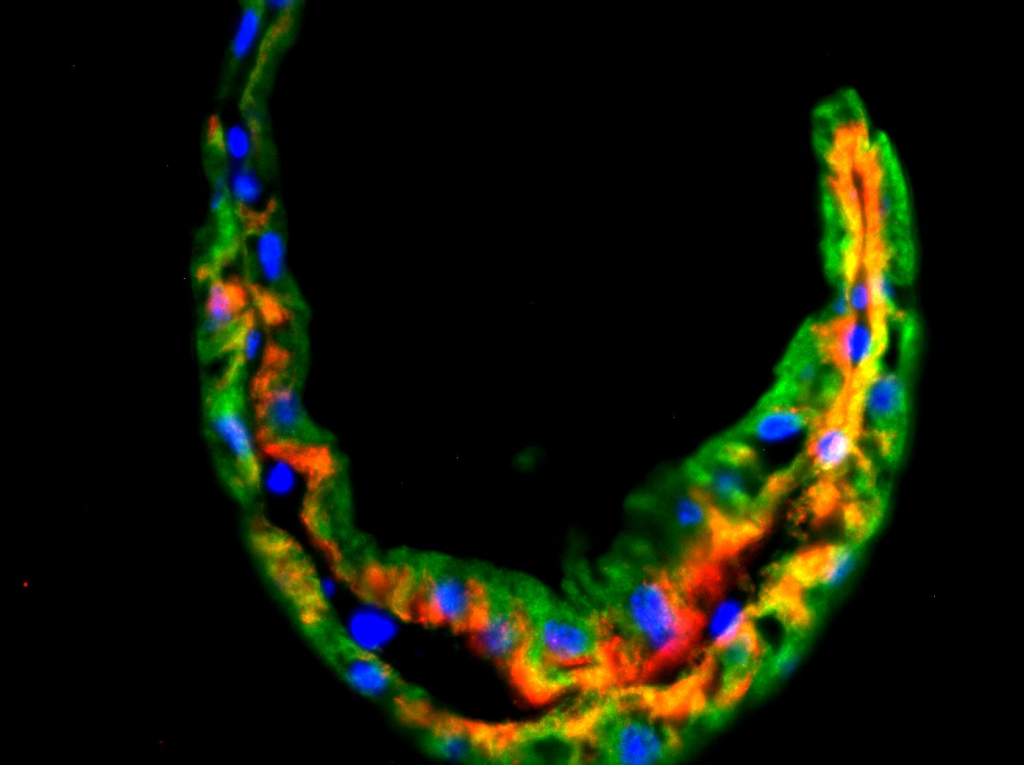
We previously showed that two major ion pumps that are batteries for ion and water movement (Na+/K+-ATPase, H+-ATPase) have a high expression in coxal gills in a manner typical of an ion-transporting epithelia, while the expression patterns in the sternal gills are different and better suited for nitrogenous waste excretion and gas transport. To develop these cellular transport models further, we are using a combination of molecular tools (bulk RNA-seq, qPCR), biochemical assays to measure enzyme activities, and electrophysiology and pharmacological inhibitors to measure ion and oxygen fluxes (e.g., scanning ion-selective electrode technique).We are also working to compare general gill structure and function across many freshwater and marine amphipod taxa using electron and confocal microscopy. Our major goals are to assess the contribution of coxal and sternal gills in adverse outcomes to salinity and low oxygen (hypoxia) challenge that is typical in polluted freshwater systems.
We hope that this research can be used to better understand the resilience of these environmentally significant animals.
